


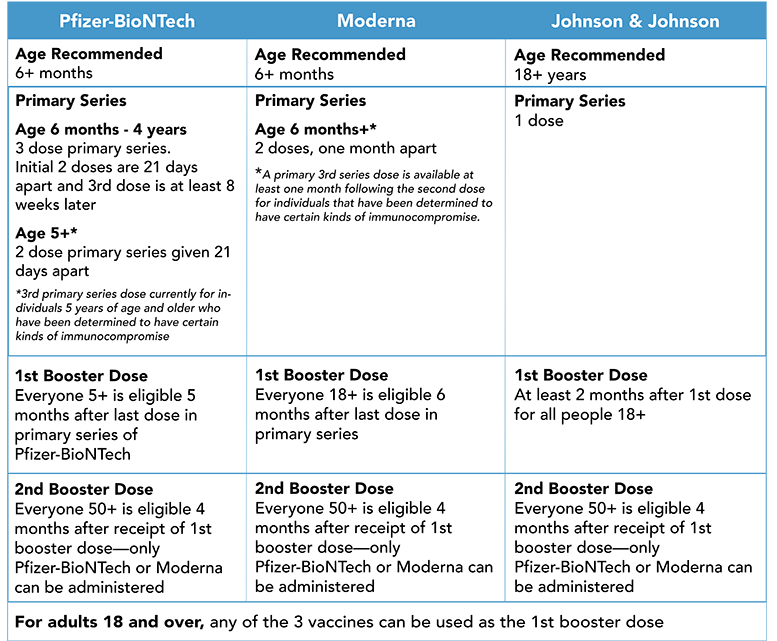
FDA authorizes Moderna and Pfizer-BioNTech COVID-19 vaccine to include children as young as six months old
On June 17, 2022, the Moderna and Pfizer-BioNTech vaccine were authorized for young children, marking a day that any parents, caregivers, and clinicians have been waiting for since the vaccines were originally introduced. The FDA amended the emergency use authorization (EUA) of the Moderna and Pfizer-BioNTech COVID-19 vaccines to include children as young as months old. There is no preference for the Moderna or Pfizer-BioNTech vaccine in this age group.
Click here for additional recommendations for immunocompromised people
View Previous Updates

